Building a culture of safety, one step at a time
In today’s rapidly evolving food ecosystem, food safety is not a single milestone — it is a continuous journey. With shifting regulations, rising consumer expectations, expanding supply chains, and increasing audit scrutiny, food businesses must adopt a proactive, improvement-oriented mindset.
A structured roadmap not only strengthens compliance but also enhances product quality, reduces risk, and builds long-term customer trust. Whether you operate a small manufacturing unit or a large-scale processing facility, a continuous improvement system helps your team stay aligned and accountable.
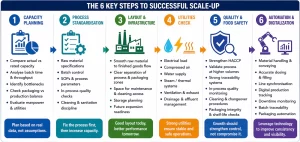
Below is a comprehensive, practical roadmap widely used across the global food industry to build and sustain world-class food safety systems.
1. Conduct a Baseline Assessment
Before improvements can begin, it’s essential to know your starting point.
A detailed baseline assessment provides clarity on what is working well and where attention is needed.
Key evaluation areas include:
- Hygiene and sanitation compliance across all production zones
- Supplier control and raw material quality consistency
- Critical Control Points (CCPs) and Operational Prerequisite Programs (OPRPs) within the HACCP plan
- Documentation practices, including SOPs, logs, and monitoring records
- Training levels and food safety culture across the workforce
A transparent gap analysis creates a strong foundation for targeted improvements rather than guesswork.
2. Plan: Set Clear Food Safety Goals
The planning stage is where strategy meets structure.
Using the PDCA (Plan–Do–Check–Act) methodology ensures systematic and measurable progress.
Effective planning involves:
- Specific, measurable goals
Example: “Reduce CCP deviations by 20% in the next quarter.” - Resource allocation, including manpower, training, equipment, and budget
- Risk-based prioritization, focusing first on high-impact areas
- Training requirements mapped to current gaps
- Compliance timelines aligned with audits and regulatory expectations
A well-crafted plan reduces reactive firefighting and encourages smooth, predictable operations.
3. Implement Improvements (DO Phase)
With a plan ready, it’s time to act.
This step translates strategy into real operational changes.
Common implementation activities include:
- Updating or refining hygiene and sanitation procedures
- Improving documentation, record-keeping, and traceability
- Conducting regular training on GMP, allergen control, CCP monitoring, and HACCP fundamentals
- Upgrading equipment, measurement tools, or automation systems
- Strengthening supplier audits, verification, and qualification processes
Even small improvements — when applied consistently — can significantly elevate the overall food safety performance of your facility.
4. Monitor, Measure & Verify
“What gets measured, gets improved.”
Continuous improvement thrives on accurate, timely data — not assumptions.
Monitoring may involve:
- CCP and OPRP verification records
- Temperature monitoring logs
- Swab results and microbiological test reports
- Internal audit findings and corrective action records
- Customer complaint trends
- Non-conformance (NC) occurrence patterns
Verification activities, such as internal audits, sampling plans, and data reviews, ensure that systems are not only implemented but functioning effectively.
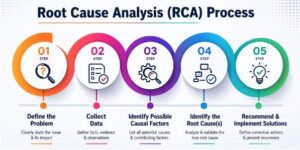
5. Analyze & Identify Gaps (CHECK Phase)
This stage is the heartbeat of continuous improvement.
You evaluate performance, identify deviations, and understand the root causes.
Useful tools include:
- Trend analysis to identify recurring issues
- Root Cause Analysis (RCA) using methods like 5 Why’s or Fishbone Diagram
- Pareto analysis to determine which issues create the most impact
- Review of audit findings, complaint patterns, and process failures
Understanding problems deeply leads to meaningful, long-term corrections rather than temporary fixes.
6. Implement Corrective & Preventive Actions (CAPA)
Once the gaps are analyzed, strong foundations must be built to prevent recurrence.
A robust CAPA system ensures:
- Immediate corrective action to fix the problem at its root
- Preventive measures to reduce the likelihood of future issues
- Revised procedures and strengthened controls
- Continuous skill-building, especially for staff performing critical tasks
- Improved monitoring systems for sensitive processes
The objective is simple: eliminate repeated non-conformities and protect the integrity of your food safety system.
7. Review, Update & Improve (ACT Phase)
Continuous improvement means regularly refreshing and re-aligning your system.
Key actions include:
- Reviewing SOPs, policies, and checklists every 6–12 months
- Updating processes based on audit findings, new risks, or regulatory changes
- Enhancing training modules to reflect new learnings
- Re-evaluating supplier performance using updated scorecards
- Integrating updates from FSSAI, CODEX, ISO, and other standards
This ensures your system evolves instead of becoming outdated.
8. Build a Strong Food Safety Culture
A system is only as strong as the people who run it.
True continuous improvement happens when the workforce believes in the importance of food safety.
To build a strong culture:
- Provide regular training & refresher programs
- Encourage employees to report near misses and unsafe practices
- Recognize and reward good food safety behavior
- Ensure leadership involvement, visibility, and accountability
- Create an environment where food safety is seen as everyone’s responsibility
Culture is the glue that holds all food safety systems together.