Food safety is one of the most critical pillars of modern food manufacturing. Across categories, from ready-to-eat foods to beverages, snacks, bakery and ingredients; manufacturers operate under increasing pressure from regulations, audits, retail expectations and consumer scrutiny. Despite having SOPs and certifications in place, many plants still experience silent safety gaps: small operational lapses that compound into contamination risks, inconsistencies and costly deviations.
Bridging these gaps requires interventions that are practical, repeatable and scientifically grounded. It also demands a holistic view of people, processes, equipment, environment and documentation. The goal is not just meeting compliance, but building a resilient, future-proof safety system.
Common Food Safety Gaps in Food Manufacturing
Food manufacturing plants often run with structured SOPs and certifications, yet hidden operational gaps continue to weaken safety systems. These gaps usually emerge during audits, deviation investigations or when production stress increases. Some of the most common include:
- Inconsistent GMP behaviour where handwashing, PPE and hygiene steps fluctuate across shifts, especially during peak loads.
- Sanitation lapses caused by rushed cleaning, incomplete disassembly or lack of verification testing.
- Weak raw material control when incoming ingredients are accepted without complete checks or supplier inconsistencies slip through.
- Allergen cross-contact risks during changeovers, shared equipment or poor segregation.
- Documentation and traceability gaps due to manual recording, illegible entries or missing batch-stage links.
- Irregular environmental monitoring where swabbing schedules are not risk based or results are not analysed for trends.
These gaps reduce audit readiness, increase variability and expose products to contamination.
Root Causes Behind These Gaps
Food safety gaps rarely originate from one incident. They stem from deeper operational constraints, cultural patterns and system-level weaknesses. Understanding the root causes helps manufacturers build stronger and more sustainable controls.
- Limited staff training where employees know the task but not the reason behind it, leading to shortcuts.
- Manual-heavy processes that create blind spots, backlogs and inconsistent data.
- Unclear or outdated SOPs that do not reflect actual plant conditions, causing confusion on the floor.
- Aging infrastructure or poor design that makes cleaning difficult and increases cross-traffic.
- Variable supplier quality that introduces microbiological, chemical or physical risks.
- Weak supervision culture where deviations are not corrected quickly or feedback loops are missing.
When these root causes are addressed, food safety systems become more consistent, predictable and resilient.
Practical Interventions to Address These Gaps
Below are the most effective interventions food manufacturers can implement today to strengthen safety, enhance quality assurance and improve audit readiness.
- Strengthen GMP Implementation & Hygiene Discipline
Good Manufacturing Practices (GMP) are the foundation of food safety, but their strength lies in everyday execution—not the manual. Many gaps arise from inconsistent hygiene behaviour, unclear SOPs or inadequate supervision.
Practical Interventions
- Introduce structured GMP patrols to reinforce discipline across handwashing, PPE usage, housekeeping and sanitation.
- Use visual SOPs at workstations so that instructions are easy to follow irrespective of language barriers.
- Create zoning controls to separate raw and finished product movement, personnel flow and equipment cleaning areas.
- Install foot-operated sanitisers, handwash counters and controlled entry hygiene stations.
These measures help standardise behaviour, reduce variability in hygiene practices and form a strong first line of defence against contamination.
- Upgrade Cleaning & Sanitation Systems
Sanitation protocols determine whether equipment surfaces remain free of pathogens, biofilms and residual allergens. Many plants rely on fixed, time-based cleaning cycles, which often do not reflect actual production loads or risk levels.
Practical Interventions
- Align cleaning schedules with production intensity, product type and contamination risk.
- Use ATP swab testing to verify whether cleaning has been effective before restarting production.
- Adopt colour-coded tools to prevent cross-area contamination.
- Strengthen CIP (Clean-in-Place) validation through temperature checks, chemical concentration monitoring and flow pattern optimisation.
- Maintain a sanitiser concentration log using testing strips to avoid over- or under-dosing.
Improved sanitation not only strengthens safety but also supports longer shelf life and consistent product quality.
- Improve Supplier & Raw Material Control
Manufacturers often underestimate the role of suppliers in food safety. Raw materials can bring microbiological, chemical or physical risks into the facility, making supplier control indispensable.
Practical Interventions
- Create a robust approved vendor program based on certifications, audit scores and historical performance.
- Implement incoming material inspection SOPs with batch sampling, documentation checks and clear rejection criteria.
- Introduce digital tracking of inbound raw materials using QR codes or batch receipt logs.
- Define cold chain unloading practices for temperature-sensitive ingredients.
- Maintain supplier scorecards to track compliance and drive continuous improvement.
Stronger raw material control reduces variability and enhances overall process safety.
- Establish a Robust Allergen Management System
Allergen contamination is one of the most common causes of recalls globally. Even small lapses—such as using the wrong scoop or switching products without proper clearance—can lead to significant risk.
Practical Interventions
- Map allergens across every ingredient, storage area and process step.
- Use dedicated colour-coded equipment for allergenic materials.
- Follow production sequencing that flows from non-allergen to allergen-containing items.
- Maintain separate storage zones for allergen ingredients.
- Mandate line clearance before changing SKUs to avoid cross-contact.
A structured allergen management program helps manufacturers reduce risk and build consumer trust.
- Adopt Technology for Digital Traceability & Real-Time Monitoring
Manual records often create blind spots. Digital systems increase visibility, control and accountability across the manufacturing cycle.
Practical Interventions
- Shift to digital batch records to improve traceability and reduce human error.
- Install IoT sensors for monitoring temperature, humidity and other critical parameters in real time.
- Implement barcode or QR-based traceability from raw materials to finished products.
- Move SOPs, MSDS and sanitation logs to a cloud-based document control system.
- Enable automated alerts for deviations such as cold storage temperature excursions.
Digitalisation supports rapid root-cause analysis, audit readiness and regulatory compliance.
- Reinforce Employee Training & Behaviour-Based Safety
Even the most advanced equipment cannot offset poor human behaviour. Food safety culture strengthens when employees understand why safety matters, not just how to follow rules.
Practical Interventions
- Conduct short, monthly micro-trainings instead of long annual sessions.
- Use demonstration-led training to reinforce correct practices.
- Introduce staff scoreboards for hygiene, housekeeping and discipline.
- Ensure supervisors maintain behaviour observation sheets to identify patterns.
- Implement a buddy system where experienced workers guide new employees.
Consistent, engaging training helps create a proactive safety culture rather than a reactive one.
- Expand & Strengthen Environmental Monitoring Programs (EMP)
Environmental monitoring reveals microbial hotspots on equipment, drains, air systems and high-touch surfaces. A strong EMP identifies contamination risks before they reach the product.
Practical Interventions
- Divide the facility into risk-based zones—raw, semi-processed and high-care (ready-to-eat).
- Set a defined swabbing schedule for drains, conveyors, tables and air handling units.
- Document and analyse test results to identify recurring patterns.
- Establish clear corrective action workflows for microbiological deviations.
- Review EMP results during internal audits and management review meetings.
EMP acts as an early-warning system, allowing manufacturers to intervene before contamination spreads.
- Optimise Facility & Equipment Design
Design plays a crucial role in preventing contamination. Poor facility layout increases cross-traffic, complicates cleaning and reduces hygiene control.
Practical Interventions
- Shift towards hygienic design equipment with smooth surfaces, welded joints and minimal dead zones.
- Ensure unidirectional flow of materials—from raw to processed to finished goods.
- Use air pressure differentials and controlled access to protect high-care areas.
- Install non-porous floors and walls that are easy to clean and resistant to wear.
- Create segregated waste disposal pathways away from product areas.
Smart facility design reduces risk, supports efficient cleaning and improves operational flow.
- Strengthen Quality Assurance, Documentation & Internal Audits
Documentation is the backbone of compliance. Many plants operate well but fail audits because they cannot prove their processes through records.
Practical Interventions
- Maintain a master document control system, preferably digital.
- Perform internal audits at planned intervals to verify safety systems.
- Record critical control parameters in real time instead of end-of-shift entries.
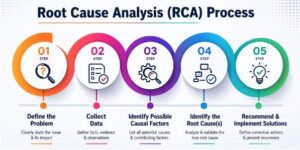
- Use structured root cause analysis (RCA) methods for non-conformances.
- Track CAPA (Corrective and Preventive Actions) to ensure gaps are addressed.
Strong documentation supports accountability, traceability and regulatory alignment.
- Validate Critical Control Points & Safety Systems
Validation ensures that safety controls perform as expected under real conditions. Without validation, CCPs (Critical Control Points) may give a false sense of security.
Practical Interventions
- Validate thermal processes through time–temperature studies.
- Conduct performance checks for metal detectors, sieves and magnets.
- Review CCP monitoring logs during every shift.
- Calibrate instruments regularly to maintain accuracy.
- Maintain deviation logs with escalation procedures for anomalies.
Validation builds confidence that every batch is produced under safe conditions.
How Beyzon Foodtek Helps Manufacturers Close Food Safety Gaps
Beyzon Foodtek partners with food and beverage manufacturers to strengthen safety and quality systems across the entire production lifecycle. We begin with a comprehensive assessment to identify food safety gaps and then design practical interventions tailored to the client’s operations and regulatory needs. Our services integrate GMP/GHP audits, HACCP/FSMS frameworks, sanitation protocols, layout optimisation, and supplier evaluation to create robust, audit‑ready systems. By embedding food safety into plant design, mechanisation, automation, and operations excellence, we make compliance actionable and sustainable. With documentation support, and continuous performance tracking, we help manufacturers reduce contamination risks, improve consistency, and operate safer, smarter facilities.
Conclusion
Food safety gaps do not disappear with paperwork—they disappear with practical, daily discipline supported by the right systems and tools. By strengthening sanitation, improving supplier control, enforcing allergen management, expanding EMPs, modernising layouts and embracing digital traceability, manufacturers can unlock a safer, more compliant and more resilient operation.
As food businesses scale, these interventions become essential not only for compliance but also for brand reputation and consumer trust. With structured support from experts like Beyzon Foodtek, manufacturers can build safety systems that withstand audits, enhance quality and support long-term growth.
FAQs
1. What are the most common food safety gaps in manufacturing?
Common gaps include poor GMP compliance, weak sanitation, inadequate raw material checks, allergen risks and incomplete documentation.
2. Why do food safety issues occur despite SOPs?
Because SOPs are often not followed consistently due to manual processes, poor training, or lack of monitoring.
3. How can sanitation practices be improved?
By using risk-based cleaning schedules, ATP testing, proper chemical control and regular hygiene audits.
4. How does digital traceability help food safety?
It improves visibility, reduces human error, strengthens recall readiness and supports audit compliance.
5. How does Beyzon Foodtek support food manufacturers?
Beyzon Foodtek helps close safety gaps through audits, GMP systems, sanitation planning, digital tracking and compliance-driven process improvement.